Intended for U.S. Healthcare Professionals
Expecting Moms
your key to
cervical ripening
Keep control at your fingertips with easy insertion and removal of CERVIDIL1
See How To Properly Administer
Proper Administration of CERVIDIL will help optimize drug delivery1
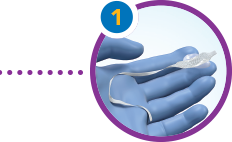
Pick up the insert between 2 fingers and lightly coat with water-miscible lubricant.
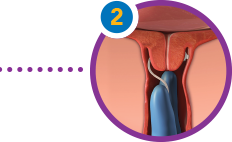
Gently place your fingers with the insert into the vagina. Position the insert transversely in the posterior vaginal fornix. Take care not to dislodge the insert when removing fingers. Slightly tuck the retrieval tape into the vagina.
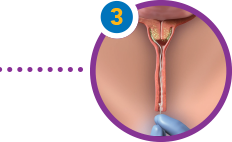
To retrieve, locate the retrieval tape and pull it gently until the product is fully removed.
Upon removal of CERVIDIL, it is essential to ensure that the slab has been removed, as it will continue delivering the active ingredient.
Important
Guidelines
- CERVIDIL must be kept frozen until use and is stable when stored in a freezer for a period of 3 years
- There is no need for previous warming of the product
- Insert immediately after removal from its foil package
- CERVIDIL does not require sterile conditions
- CERVIDIL must not be used without its retrieval system
- Do not wrap the retrieval tape around the insert before insertion
- Do not overlubricate; excess lubricant could prevent optimal swelling and release of dinoprostone from the vaginal insert
- Patients should remain in the recumbent position for 2 hours following insertion, but thereafter may be ambulatory

Just One Dose
A single dose to help ripen the cervix1
- Controlled-release drug delivery – Formulated to release 0.3 mg/hr for up to 12 hours
- Releases drug at the cervix
- Up to 12 hours of drug delivery without multiple administrations
Removed In Seconds
Cleared In Minutes
Maintain control and
retrieve with ease1
- Long tape facilitates immediate removal
- The half-life of dinoprostone is 2.5 to 5 minutes
When to remove CERVIDIL
- 12 hours after insertion or upon onset of active labor, or whichever comes first
- Prior to amniotomy
- With any evidence of uterine hyperstimulation, sustained uterine contractions, fetal distress, and other fetal or maternal adverse reactions
- Before oxytocin administration is initiated; the patient’s uterine activity should be carefully monitored for uterine hyperstimulation


