Induction and cervical dilation

Your delivery starts
with your cervix
A relaxed and open cervix is the
first step in delivering your baby.
During pregnancy, your cervix plays the role of keeping your baby in your womb. For labor to begin, your cervix needs to be open (dilated) and relaxed. If your cervix isn’t ready, all the pushing in the world won’t help your baby come out.
Here's how it works
Thin
As labor begins, your cervix will
change from being as thick as
your nose to being as thin as a
piece of paper.
Soften
For months your cervix has
been firm, but now needs to
become soft and stretchy.
Open [Dilate]
While your cervix thins and softens,
it may also begin to dilate. Your cervix
will continue to open throughout
labor due to contractions.
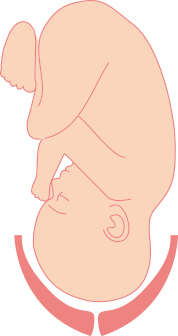
Cervix During Pregnancy
Cervix is closed, thick and hard to protect the baby
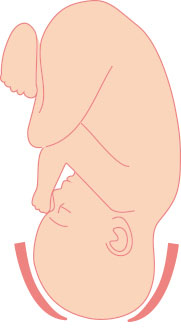
Cervix during Labor
Cervix thin, softened, and open [dilated] so labor can begin
After your cervix is relaxed and dilated, you’ll be ready for the next step. This is when regular labor contractions will begin to help push your baby out.
My cervix may need a pep talk.
It’s showtime for your cervix
While you may be more than ready to start labor, your cervix might have other plans. Fortunately, CERVIDIL may be able to assist you (and your cervix) to keep things moving. Read on to learn more about the role your cervix plays as your due date draws near.
Checking on your Cervix
With regard to your cervix, you may feel a twinge from time to time, but it is entirely possible that you may not feel anything at all. The only way to know if your cervix is relaxed and dilated is for your doctor or midwife to check.
Only time will tell
Your cervix is on its own schedule when it comes to relaxation and dilation. All women are different, and it can be difficult to predict when cervical relaxation begins. It can begin at any time—a few weeks before your due date, the night before, or not at all. If very little or nothing has happened with your cervix, your doctor may recommend something to help, like CERVIDIL.
CERVIDIL may help
CERVIDIL is a vaginal insert specifically designed to help relax and open your cervix.
CERVIDIL is the only
FDA-approved vaginal insert with time-released medication that may help your cervix gradually soften, thin, and dilate.
Why does that matter? Because your cervix needs to open in order for you to have a vaginal delivery. Explore more and see how CERVIDIL may assist you during induction.
